What Is The Oxidation State Of Hg In Hg2Cl2
What Is The Oxidation State Of Hg In Hg2Cl2 - Your solution’s ready to go! Each hg atom has an oxidation state of +1 in this compound. The oxidation state of hg in hg2cl2 is +1. The oxidation state of sn is +2 since the oxidation number of hg increased it is oxidized and since the oxidation number of sn is reduced it is reduced. The oxidation state of hg in hg2cl2 is +1. What is the oxidation state of hg in hg2cl2? The oxidation state of hg in hg2cl2 is +1.
The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1. What is the oxidation state of hg in hg2cl2? Your solution’s ready to go! Each hg atom has an oxidation state of +1 in this compound. The oxidation state of hg in hg2cl2 is +1. The oxidation state of sn is +2 since the oxidation number of hg increased it is oxidized and since the oxidation number of sn is reduced it is reduced.
The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1. What is the oxidation state of hg in hg2cl2? Your solution’s ready to go! Each hg atom has an oxidation state of +1 in this compound. The oxidation state of sn is +2 since the oxidation number of hg increased it is oxidized and since the oxidation number of sn is reduced it is reduced.
An impure sample of sodium oxalate `(Na_(2)C_(2)O_(4)` weighing 0.20 g
The oxidation state of hg in hg2cl2 is +1. Each hg atom has an oxidation state of +1 in this compound. Your solution’s ready to go! The oxidation state of sn is +2 since the oxidation number of hg increased it is oxidized and since the oxidation number of sn is reduced it is reduced. The oxidation state of hg.
Oxidation state of hydrogen in CaH2 is (1) +1 (2) −1 Filo
The oxidation state of hg in hg2cl2 is +1. Your solution’s ready to go! Each hg atom has an oxidation state of +1 in this compound. The oxidation state of sn is +2 since the oxidation number of hg increased it is oxidized and since the oxidation number of sn is reduced it is reduced. What is the oxidation state.
How to find the Oxidation Number for Hg in Hg2Cl2 YouTube
Each hg atom has an oxidation state of +1 in this compound. The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1. What is the oxidation state of hg in hg2cl2?
SOLVED What is the oxidation number of Hg in Hg2Cl2? a) 2 b) 0 c) 1
The oxidation state of hg in hg2cl2 is +1. Your solution’s ready to go! The oxidation state of hg in hg2cl2 is +1. Each hg atom has an oxidation state of +1 in this compound. The oxidation state of sn is +2 since the oxidation number of hg increased it is oxidized and since the oxidation number of sn is.
Question Video Deducing the Oxidation State of Oxygen in Hydrogen
The oxidation state of hg in hg2cl2 is +1. The oxidation state of sn is +2 since the oxidation number of hg increased it is oxidized and since the oxidation number of sn is reduced it is reduced. What is the oxidation state of hg in hg2cl2? Each hg atom has an oxidation state of +1 in this compound. Your.
[SolveLancer Test] With reference to aqua... Physical Chemistry
The oxidation state of hg in hg2cl2 is +1. Each hg atom has an oxidation state of +1 in this compound. The oxidation state of hg in hg2cl2 is +1. What is the oxidation state of hg in hg2cl2? Your solution’s ready to go!
How to Assign Oxidation Numbers
Your solution’s ready to go! The oxidation state of hg in hg2cl2 is +1. What is the oxidation state of hg in hg2cl2? The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1.
SOLVED 9. Assign the oxidation numbers of each element given in bold
The oxidation state of hg in hg2cl2 is +1. Each hg atom has an oxidation state of +1 in this compound. The oxidation state of hg in hg2cl2 is +1. What is the oxidation state of hg in hg2cl2? The oxidation state of hg in hg2cl2 is +1.
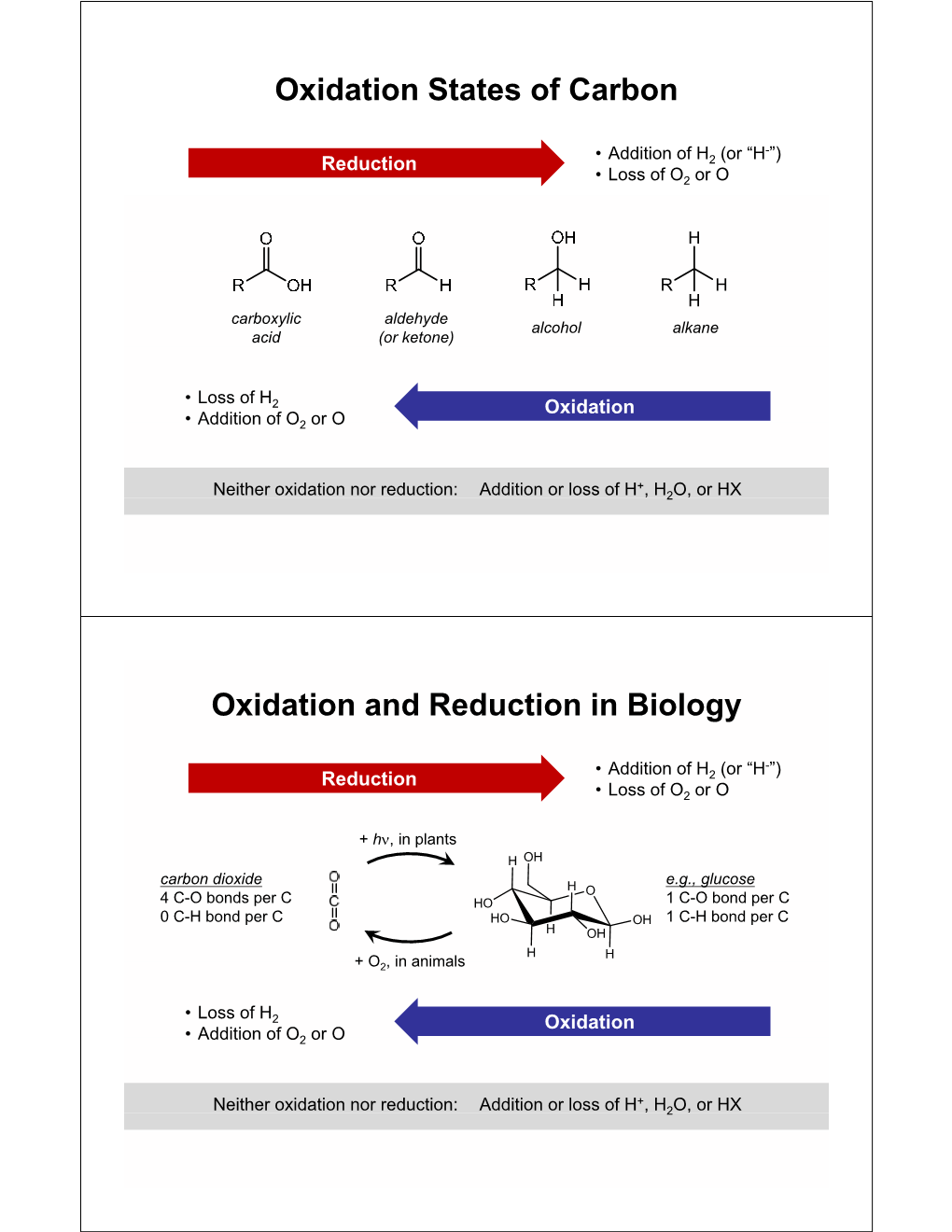
Oxidation States of Carbon Oxidation and Reduction in Biology DocsLib
The oxidation state of sn is +2 since the oxidation number of hg increased it is oxidized and since the oxidation number of sn is reduced it is reduced. The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1. What is the oxidation.
The Oxidation State Of Hg In Hg2Cl2 Is +1.
The oxidation state of hg in hg2cl2 is +1. The oxidation state of hg in hg2cl2 is +1. What is the oxidation state of hg in hg2cl2? Each hg atom has an oxidation state of +1 in this compound.
The Oxidation State Of Sn Is +2 Since The Oxidation Number Of Hg Increased It Is Oxidized And Since The Oxidation Number Of Sn Is Reduced It Is Reduced.
Your solution’s ready to go!





![[SolveLancer Test] With reference to aqua... Physical Chemistry](https://media.kunduz.com/media/question/raw/20210620074505769111-3084283.jpg?h=512)